- Blog
- Flash ecu motorcycle
- Download driver asus smart gesture windows 10
- Professional resume template microsoft word
- Chemistry periodic table with names of elements
- Real estate flyer design clipart
- Single letter tattoo fonts
- Hindi typing keyboard kruti dev chart
- Vlc media player safe download for mac
- Simcity 4 rush hour no cd crack
- Logitech setpoint mouse and keyboard battery life
- Free paw patrol svg for cricut
- Minecraft twilight forest mod download 1-12-2
- Equifax not lifting my credit freeze
- Albert einstein atomic theory
- Pc games free download action
- Bass trombone slide position chart
- Sins of a solar empire v1-9 trainer
- Free printable mens ring sizer
- Among us crewmate coloring page
- Lettering fonts tattoo
- Monitor cpu gpu temps
- Minecraft 1-9 apk download
- Types of drop ceiling grid
- Personal budget planning template
- Project cost planner in excel
- California drivers license template editable word
- Free online pdf editor download
- Games pinball 3d
- Windows 11 fresh install
- Mmpi 2 test questions pdf
- Microsoft access sample database orders relationship diagram
- Klondike solitaire green felt turn 3 game
- Hentai bondage gag blindfold sensory deprivation
- Preschool sight words worksheets
- Free jailbreak vip server 2019
- Norton ghost 2003 iso
- Google doc template for avery label 5160
- Multiplication timed time table worksheet
- Youtube downloader mp3 online free converter
- Minecraft windows 10 edition inventory hack 1-12
- Spectrum tv choice changing channels
- Structural functionalism difference from conflict theory
- Free crm software download small business
- Big minion butt
- Android install for apple ios free download
- Gary arbuckle married to amy freeze
- Moca medical test scoring

Please report any accidental mistake in the above statistics on chemical elements. There you can find the metals, semi-conductor(s), non-metal(s), inert noble gas(ses), Halogens, Lanthanoides, Actinoids (rare earth elements) and transition metals.Ĭlick here: for a schematic overview of the periodic table of elements in chart formĭo you need to know the weight of some molecules? Try our Molecular Weight Calculator! Handouts can provide only necessary information during test taking. Hide unnecessary or grade-level inappropriate information. Please note that the elements do not show their natural relation towards each other as in the Periodic system. Ptables new, up-to-date periodic table PDF and wide periodic table PDF are layered so you can choose exactly what you want to print, and are the perfect companion to the periodic table classroom poster. The first chemical element is Actinium and the last is Zirconium.

The chemical elements ofįor chemistry students and teachers: The tabular chart on the right is alphabethically listed. By convention, elements are organized in the periodic table, a structure that captures important patterns in their behavior.Devised by Russian chemist Dmitri Mendeleev (18341907) in 1869, the table places elements into columnsgroupsand rowsperiodsthat share certain properties. This list contains the 118 elements of chemistry. Separation and Concentration Purification RequestĬhemical elements alphabetically listed The elements of the periodic table sorted by name in an alphabetical list.Ĭlick on any element's name for further chemical properties, environmental data or health effects.Plant Inspection & Process Optimalisation The table below consists of 118 elements of the periodic table, sorted by atomic number, atomic weight, symbols, density, discovered year and the group.(In the modern periodic table, a group or family corresponds to one vertical column. This periodic table of elements provides comprehensive data on the chemical elements including scores of properties, element names in many languages and most known nuclides (Isotopes). The periodic table allows chemists a shortcut by arranging typical elements according to their properties and putting the others into groups or families with similar chemical characteristics. Were it not for the simplification provided by this chart, students of chemistry would need to learn the properties of all 118 known elements. The term “periodic” is based on the discovery that elements show patterns in their chemical properties at certain regular intervals.
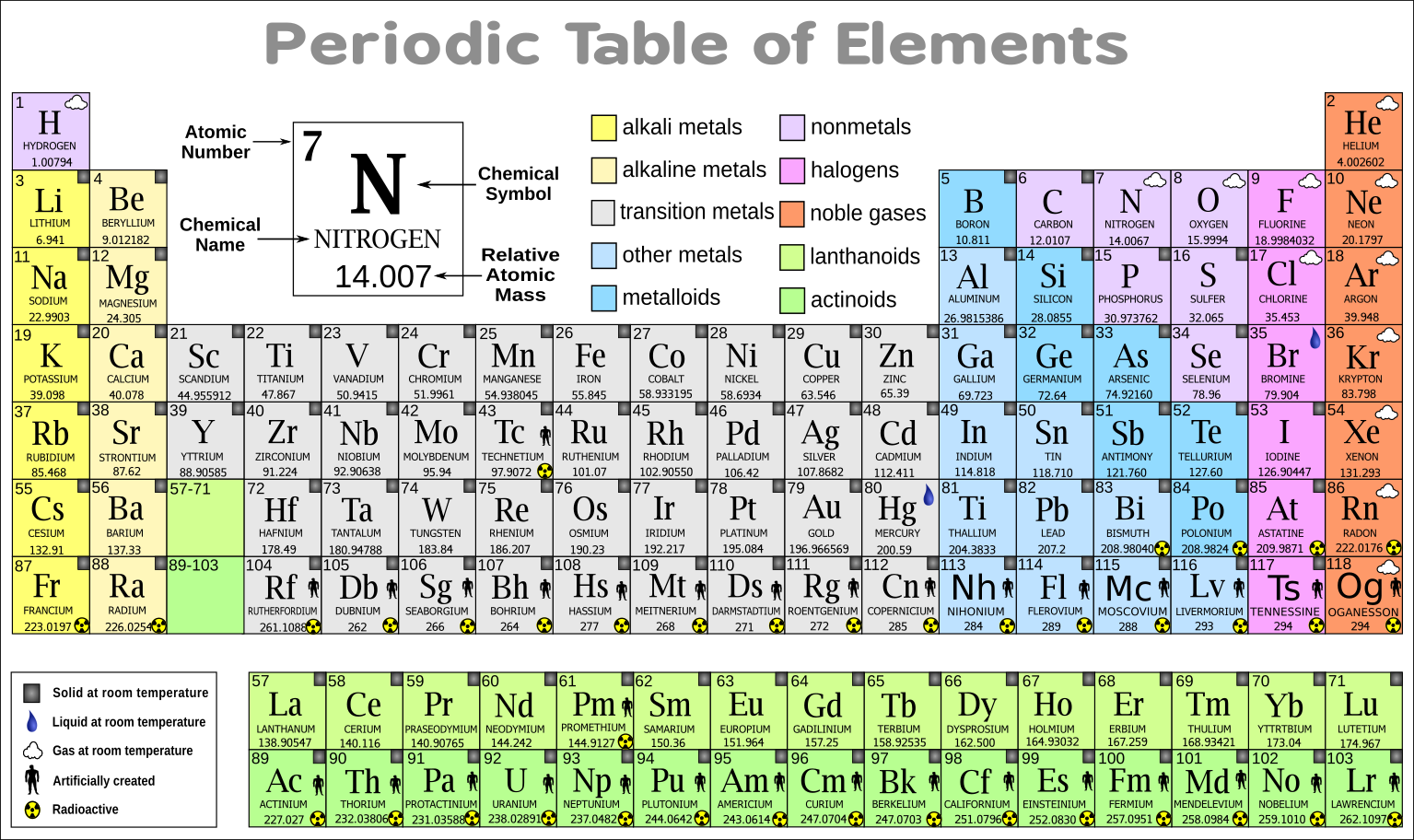
Mendeleev left spaces for elements he expected to be discovered, and today’s periodic table contains 118 elements, starting with hydrogen and ending with oganesson, a chemical element first synthesized in 2002 at the Joint Institute for Nuclear Research (JINR) in Dubna, Russia, by a team of Russian and American scientists. Its story is over 200 years old, and throughout its history, it has been a subject for debate, dispute and alteration.Īttempts to classify elements and group them in ways that explained their behavior date back to the 1700s, but the first actual periodic table is generally credited to Dmitri Ivanovich Mendeleev, a Russian chemist who in 1869 arranged 63 known elements according to their increasing atomic weight. Metals reside on the left side of the table, while non-metals reside on the right. Go into any scientist’s office or lecture hall anywhere in the world and you are likely to see one. The periodic table (also known as the periodic table of elements) is organized so scientists can quickly discern the properties of individual elements such as their mass, electron number, electron configuration and their unique chemical properties. There is no more enduring reflection of science than the Periodic Table of Chemical Elements, which sheds light not only on the essence of chemistry but physics and biology as well.
- Blog
- Flash ecu motorcycle
- Download driver asus smart gesture windows 10
- Professional resume template microsoft word
- Chemistry periodic table with names of elements
- Real estate flyer design clipart
- Single letter tattoo fonts
- Hindi typing keyboard kruti dev chart
- Vlc media player safe download for mac
- Simcity 4 rush hour no cd crack
- Logitech setpoint mouse and keyboard battery life
- Free paw patrol svg for cricut
- Minecraft twilight forest mod download 1-12-2
- Equifax not lifting my credit freeze
- Albert einstein atomic theory
- Pc games free download action
- Bass trombone slide position chart
- Sins of a solar empire v1-9 trainer
- Free printable mens ring sizer
- Among us crewmate coloring page
- Lettering fonts tattoo
- Monitor cpu gpu temps
- Minecraft 1-9 apk download
- Types of drop ceiling grid
- Personal budget planning template
- Project cost planner in excel
- California drivers license template editable word
- Free online pdf editor download
- Games pinball 3d
- Windows 11 fresh install
- Mmpi 2 test questions pdf
- Microsoft access sample database orders relationship diagram
- Klondike solitaire green felt turn 3 game
- Hentai bondage gag blindfold sensory deprivation
- Preschool sight words worksheets
- Free jailbreak vip server 2019
- Norton ghost 2003 iso
- Google doc template for avery label 5160
- Multiplication timed time table worksheet
- Youtube downloader mp3 online free converter
- Minecraft windows 10 edition inventory hack 1-12
- Spectrum tv choice changing channels
- Structural functionalism difference from conflict theory
- Free crm software download small business
- Big minion butt
- Android install for apple ios free download
- Gary arbuckle married to amy freeze
- Moca medical test scoring
